Gosuke Hayashi
Efficient Chemical Synthesis of Polyubiquitin and Ubiquitinated Proteins
 |
Gosuke Hayashi, PhDDepartment of Biomolecular Engineering, Graduate School of Engineering, Nagoya University |
|---|
Research summary
Proteins including ubiquitin are polyamide molecules, in which 20 proteinogenic amino-acids are precisely polymerized, and have been mainly prepared by genetic engineering techniques using ribosomal translation system inside living cells. However, chemical protein synthesis (CPS), in which proteins of interest are prepared through completely chemical process consisting of solid-phase peptide synthesis (SPPS) and following peptide ligation, has been significantly developed in the past two decades. Since various unnatural amino-acids can be site-specifically and multiple incorporated in SPPS procedure, CPS can produce proteins including not only modifications such as posttranslational modifications and/or functional molecules (e.g. fluorescent dyes and crosslinkers) but also unnatural structures such as D-amino acids or N-methyl amino acids.
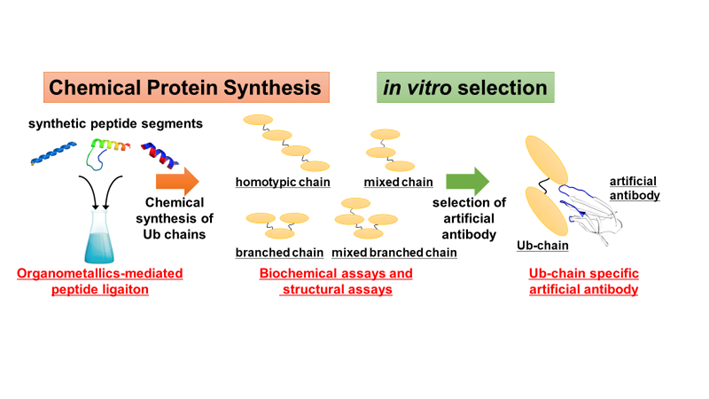
Our research group has been engaged in developing novel strategies for CPS and applying chemically synthesized proteins into molecular biology researches. For example, we have developed a new one-pot peptide ligation method utilizing organometallic complexes and DNA scaffold, and a new thioester precursors that can be used in native chemical ligation (NCL), which is one of the most popular peptide ligation methods. Furthermore, posttranslationally-modified histone proteins or fluorescently-labeled histone proteins have been synthesized and used in functional and structural analyses. In this ubiquitin project, we planned to develop a novel method to efficiently produce polyubiquitin chains or ubiquitinated proteins. Now, we are working on several collaborating projects such as chemical synthesis of ubiquitinated proteins, new methodology for ubiquitin proteomics, and artificial antibodies binding to ubiquitin or ubiquitinated proteins.
Publications
- Yanase M, Nakatsu K, Cardos C J, Konda Y, *Hayashi G, *Okamoto A.
Cysteinylprolyl Imide (CPI) Peptide: A Highly Reactive and Easily Accessible Crypto-thioester for Chemical Protein Synthesis.
Chem. Sci. 10, 5967-5975 (2019)
PMID: 31360403 - Kamo N, *Hayashi G, *Okamoto A.
Chemical Synthesis of Cys-Containing Protein via Chemoselective Deprotection with Different Palladium Complexes.
Org. Lett. 21, 8378-8382 (2019)
PMID: 31560553 - Nakatsu K, Yanase M, *Hayashi G, *Okamoto A.
Fmoc-Compatible and C-terminal-Sequence-Independent Peptide Alkyl Thioester Formation Using Cysteinylprolyl Imide.
Org. Lett. 22, 4670-4674 (2020)
PMID: 32484687 - Kondo T, Iwatani Y, Matsuoka K, Fujino T, Umemoto S, Yokomaku Y, Ishizaki K, Kito S, Sezaki T, Hayashi G, *Murakami H.
Antibody-like proteins that capture and neutralize SARS-CoV-2.
Sci. Adv. 6, eabd3916 (2020)
PMID: 32948512 - Hao F, Murphy K. J, Kujirai T, Kamo N, Kato J, Koyama M, Okamoto A, Hayashi G, Kurumizaka H, Hayes J. J.
Acetylation-modulated communication between the H3 N-terminal tail domain and the intrinsically disordered H1 C-terminal domain.
Nucl. Acids Res. 48, 11510-11520 (2020)
PMID: 33125082 - Kondo T, Eguchi M, Kito S, Fujino T, Hayashi G, Murakami H.
cDNA TRAP display for rapid and stable in vitro selection of antibody-like proteins.
Chem. Commun. 57, 2416-2419 (2021)
PMID: 33554979 - Kamo N, Kujirai T, Kurumizaka H, Murakami H, *Hayashi G, *Okamoto A.
Organoruthenium-Catalyzed Chemical Protein Synthesis to Elucidate the Functions of Epigenetic Modifications on Heterochromatin Factors.
Chem. Sci. AOP (2021)
Former Publications
- Sueoka T, Hayashi G, *Okamoto A.
Regulation of the Stability of the Histone H2A-H2B Dimer by H2A Tyr57 Phosphorylation.
Biochemistry 56, 4767-4772 (2017)
PMID: 28813589 - Kamo N, Hayashi G, *Okamoto A.
Efficient peptide ligation between allyl-protected Asp and Cys followed by palladium-mediated deprotection.
Chem. Commun. 54, 4337-4340 (2018)
PMID: 29644365 - Sueoka T, Koyama K, Hayashi G, *Okamoto A.
Chemistry-driven Epigenetic Investigation of Histone and DNA Modifications.
Chem. Rec. 18,1727-1744 (2018)
PMID: 30070422 - Kamo N, Hayashi G, *Okamoto A.
Triple Function of 4-Mercaptophenylacetic Acid Promotes One-Pot Multiple Peptide Ligation.
Angew. Chem. Int. Ed. 57, 16533-16537 (2018)
PMID: 30346110 -
Hayashi G, Yanase M, Nakatsuka Y, *Okamoto A.
Simultaneous and Traceless Ligation of Peptide Fragments on DNA Scaffold.
Biomacromolecules 20, 1246-1253 (2019)
PMID: 30677290





